
Catalysis
Design and production of advanced novel catalytic materials.
Team
Principal Investigator
Students
- Zeping Du (MSc
- Aina Syadhida Jamal (PhD)
- Alberto Palmer Pardo (MSc)
- Antonio Torres Lopez (PhD)
- Yang Peng (PhD)
- Huaizhong Xiang (PhD)
- Yiheng Xiao (PhD)
- Xinyue Zhou (PhD)
Multi-step reactions represent a key step towards the utilisation of renewable biomass feedstocks and their derivatives for sustainable chemical production. Whilst convention has typically dictated an approach of focusing on each single step discreetly, catalytic cascades offer significant economic and environmental advantages.
Developing multifunction materials, possessing two or more different active sites, is critical to the exploitation of such processes. Single-atom catalytic sites represent the ultimate in heterogeneous catalysis active site miniaturisation, although optimal performance may not necessarily have been achieved.
The size gap between these single sites and nanoparticle systems spans from two-atom species up to clusters of tens of atoms.
This size domain may well prove to be superior, especially in bimolecular reactions or where bimetallic species have displayed interesting synergies.
Establishing these two independent but interwoven themes will stimulate operando spectroscopy developments while simultaneously utilising the advanced imaging capabilities at Diamond. The growth of these research areas will foster new collaborations, while the resulting scientific insight will provide the platform to foster greater industrial links and support funding applications.
Projects
Unlocking multi-step catalytic cascade reactions necessitates the development of novel catalytic systems to predictably drive a cascade so that each individual catalytic transformation occurs solely over the desired site.
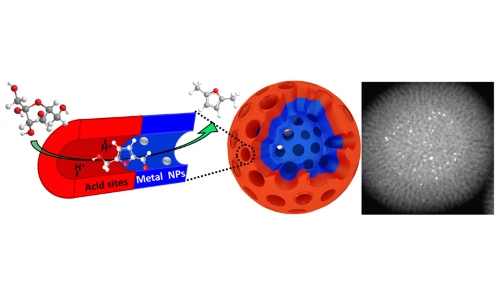

The spatial compartmentalisation of two different Platinum group metal active sites, within a hierarchical porous structure represent one strategy, with the power of such advanced materials already displayed for a cascade selective oxidation reaction.
Sub-nanometre active species – from angstrom-sized clusters to single-atom alloys represent a key driver to efficient utilisation of global resources.
Reactions on single-atom alloy systems have shown promise, eg. hydrogen dissociation and methane activation, although further synthesis developments and on-stream stability assessments are paramount if these are to find industrial applications.
Additionally, the potential advantage of diatomic and smaller cluster active species, especially with regard to bimetallic systems, is still to be explored.
